

GREDECO
Groupe de Recherche et d'Evaluation en Dermatologie et Cosmétologie
STUDY OBJECTIVE
The objective of this study conducted on 30 subjects was to clinically evaluate the anti-aging effects of photomodulation by red and infrared LED after 4 weeks of treatment.
The following clinical criteria were examined:
- Profilometry of wrinkles and fine lines - photographic camera: LifeViz Micro.
- Skin homogeneity (chromametry)
- Skin thickness and density (ultrasound)
CLINICAL PROTOCOL
Pre-selection visit
Recruitment by a dermatologist, Dr. Boisnic, on 30 healthy volunteers (Caucasian women) with fine lines and wrinkles of the crow’s feet and/or forehead, and sagging skin.
Day 0 (D0) (day of inclusion into the study):
Inclusion Criteria:
- Between 35 and 75 years of age.
- Phototype I to IV.
- Subject with fine lines and wrinkles while both relaxed and with a mobile face, lack of hydration and laxity of the skin.
- Subject who agrees to not expose herself to sun or to UV during the entire treatment duration.
Exclusion Criteria
- Subject participating in another clinical study for the evaluation of drugs or medical devices.
- Subject having received by injection or by implant non-resorbable material in the face.
- Subject with recurrent herpes on the face
- Subject treated with oral or injectable corticoids (or treatment terminated before a delay of 3 months). Note: inhaled corticoids and topical corticoids that are not applied topically on face and neck are authorized.
- Subject treated with immunosuppressive therapy or chemotherapy (or treatment terminated before a delay of 2 years).
- Subject with a history or an autoimmune medical condition or a collagen disease.
- Epileptic subject.
- Pregnant or lactating woman.
Treatment
The device used is a panel measuring 818 square centimeters (126 square inches) fitted with 192 red LED and 240 infrared LED. The wavelengths used are red (660 nm, power 26,3mW/ cm2) and infrared (850nm, power 26,6mW/ cm2). The program used is level 4, with 1 minute of infrared, 2.30 mn of red and 30 seconds of infrared, ie a total of 4 minutes.
Each subject did the treatment 2 times a week during 4 weeks (a total of 8 treatments) at GREDECO Laboratory.
After 1 treatment, the dose received by each subject is 2,4 J/ cm2 for the 850 nm wavelength (infrared), and 3,9J/ cm2 for the 660 nm wavelength (red). It was noticed that the forehead was much closer to the LED panel (0,5 cm) than the cheeks (2 cm).
Criteria for evaluating efficacy
Analysis at D0 and after 5 weeks (1 week after the last LED exposure):
- Clinical evaluation of the depth of crow’s feet and wrinkles on forehead/or neck. Measured by Lemperle dermatological scoring from 0 to 5.
-
Measure of the depth of crow’s feet and wrinkles on forehead/or neck with photographs taken by LifeViz® Micro camera and with Dermapix® software.
Results are expressed in average depth (Avg. Depth) and the unit of measure is mm. This parameter is equal to the total volume (positive volume – negative volume) divided by the surface. Measures must be taken by perfectly synchronizing the photographs in both sequences of analysis and by propagating the exact contour of the selected wrinkle at D0.
- Photographs in 2D and 3D (full face and profile) with LifeViz II macro and Life Viz micro to visualize the modulation of signs of ageing after treatment on face and/or décolleté.
-
Chromametric Analysis
Using a Konica Minolta CR/DP-400® Chromometer in order to measure the homogeneity of the skin following treatment. Two measurements were taken on the inner and outer cheek. A calculation of the difference between the two measurements was made: a reduction in the difference between these two measurements demonstrates increased homogeneity of the skin.
Measure of skin glow on the cheek measured with the L parameter of the chromameter. When the clearness of the skin increases, the L parameter increases.
- Measure of the dermis’ density and thickness on the cheek with Monaderm Ultrasound; 2D 20 Mhz probe (zone of 12.1 mm focused straight with software developed and analyzed by Advanced Control.
- Evaluation of tolerance by the subject and the dermatologist.
- Evaluation of the tolerance of the device by the subject and dermatologist.
- Self-Evaluation at each session concerning the appreciation of the device’s efficacy.
-
Statistical Analysis: An average ± SD (standard deviation) was calculated for each analyzed parameter.
The statistical analysis was made with Excel spreadsheet (version 14.4.7; 141117), using the T-Test also called standard score or paired sample test (one-tailed unequal variance) with an alpha risk set at 0.5%.
RESULTS
A total of 30 subjects were included in the study with an average age of 51.5 ± 10.3 years.
Tolerance was excellent for all 30 volunteers.
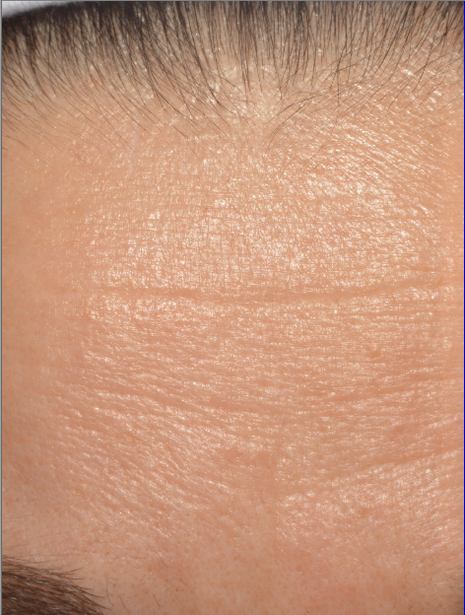
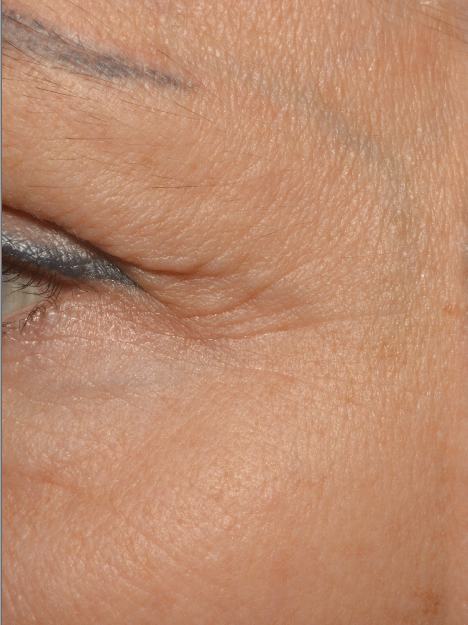
A smoothing of the skin with a decrease in the depth of crow’s feet and forehead wrinkles. This result is particularly visible on the forehead
After 8 LED treatments, the depth of forehead wrinkle measured by image analysis has significantly decreased by 32,2% for 27 subjects, with an average 0,061 mm versus 0,090 at D0 (p= 0,00053).
 |
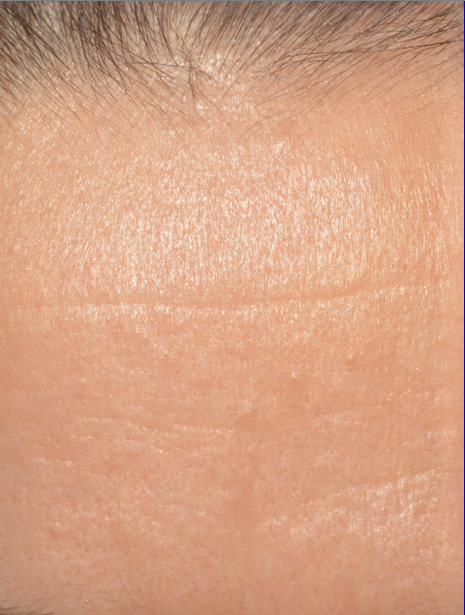 |
| Before | After 35 Days |
 |
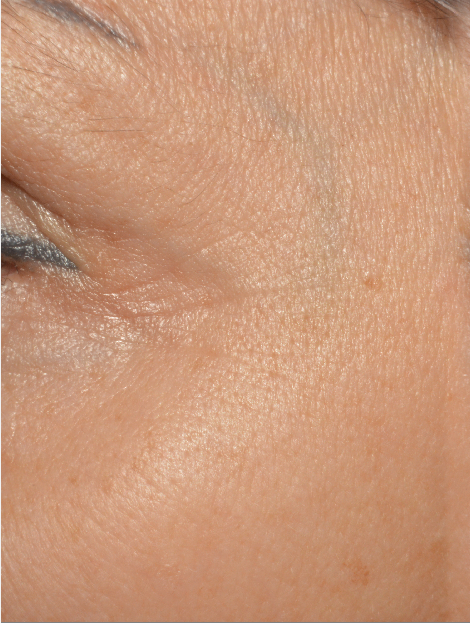 |
| Before | After 35 Days |
An increase of 6,5% of skin density was measured by ultrasound.
 |
 |
| Before | After 35 Days |
A 22% increase of skin homogeneity on the face and the décolleté
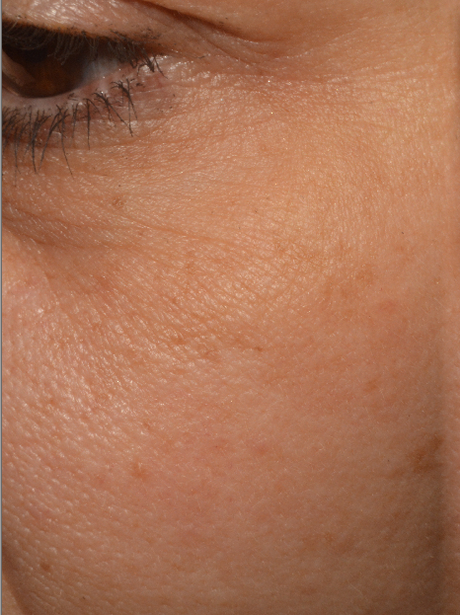 |
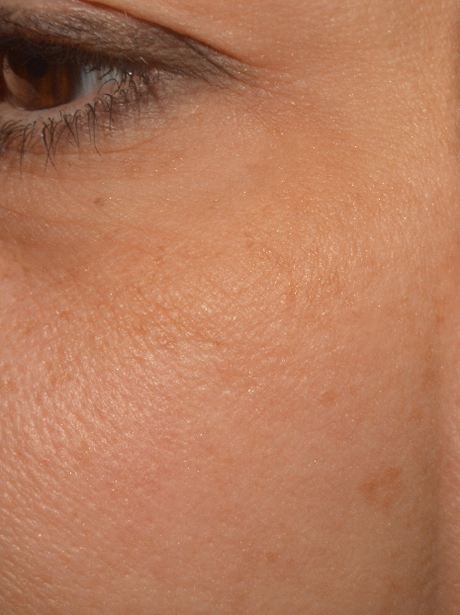 |
| Before | After 35 Days |
Conclusion
This clinical study conducted on 30 subjects experienced that after 8 treatments of photobiomodulation by LED emitting red and infrared light:
- A global rejuvenation of the face
- A 22% increase of skin homogeneity on the face and the décolleté.
- A smoothing of the skin with a decrease in the depth of crow’s feet and forehead wrinkles. This result is particularly visible on the forehead
- An increase of 6,5% of skin density was measured by ultrasound
- 67% of the subjects noticed that this treatment improved their skin and 50% are ready to purchase the device.
- Dermatological tolerance was excellent for all 30 subjects.



